views
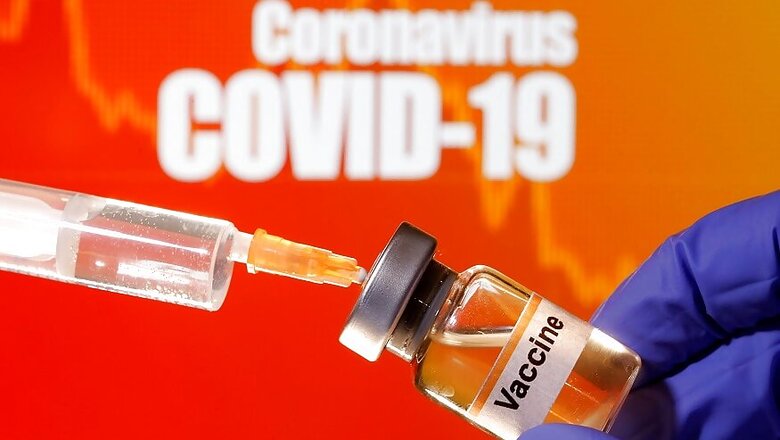
It has been a good week for positive news vis-à-vis vaccines for COVID-19 in India and outside. In India, two vaccine candidates of Zydus Cadila and Bharat Biotech entered crucial human trials phase this week.
Zydus Cadila, in an official statement today, said that its vaccine candidate, ZyCoV-D, has entered the human trials stage.
“The Adaptive Phase I/ II human clinical trials of its plasmid DNA vaccine, ZyCoV-D commenced today with the first human dosing. The Adaptive Phase I/II dose escalation, multi-centric study will assess the safety, tolerability and immunogenicity of the vaccine” the company said.
Zydus Cadila Chairman Pankaj Patel said, “This is an all important step in our fight against COVID-19.” ZyCoV-D was found to be safe, immunogenic, well tolerated in the pre-clinical toxicity studies, says Zydus Cadila. The vaccine was able to elicit high level of neutralising antibodies in animal studies.
On Tuesday, Director General of ICMR, Professor Balram Bhargava, said, “It is a moral duty to develop them as fast as possible because more than half a million people have succumbed to the disease across the world. So, fast-tracking these vaccine becomes very important.”
He was referring to two Indian vaccine candidates that have undergone successful toxicity studies in rats, mice and rabbits. Bharat Biotech has started human trials on its vaccine candidate.
Covaxin is being developed by the Hyderabad-based biotech company and Indian Council of Medical Research (ICMR) and is India’s first indigenous COVID-19 vaccine. Zydus Cadila's ZyCoV-D is the second.
Another significant development was Serum Institute's Oxford University's vaccine candidate being sent the Central Drugs Laboratory in Kasauli to determine the sterility.
It takes at least 14 days to test sterility, which is the first step critical to patient safety and a deciding factor on whether the vaccine candidate is safe for human dosage. A source at Serum Institute said, “Expect good news in August” on a question on how soon will the human trials start.
Significantly, on Wednesday, US Biotech firm Moderna Inc's experimental vaccine for COVID-19 showed it was safe and importantly provoking immune response.
“mRNA-1273 was generally safe and well-tolerated, with no serious adverse events reported through Day 57. Adverse events (AEs) were generally transient and mild to moderate in severity,” Moderna said in a statement. Moderna is in its phase 2 trial now, and will be starting phase three on July 27.
Interestingly, President Donald Trump tweeted ' Great News On Vaccines', presumably referring to Moderna but possibly even to news of British Pharmaceutical Company AstraZeneca's Vaccine candidate.
"Positive news on initial trials of the University of Oxford’s potential COVID-19 vaccine that has been licensed to AstraZeneca could be announced as soon as Thursday" reported the ITV network. This is a vaccine which is already in Phase 3 trial stage. The data are expected to be published by The Lancet medical journal.
The World Health Organisation has identified the Covid-19 vaccine candidates developed by Oxford University-AstraZeneca and Moderna Inc as the front-runners.


















Comments
0 comment